How Do Catalytic Converters Work?
Posted: January 3, 2023
Understanding how to scrap catalytic converters is a feat, but understanding how they work when connected to your car is a completely different challenge. It’s hard to imagine that your car has all these expensive precious metals inside this steel can that helps with the fumes emitted from your car. But that is what a catalytic converter is. Below is a simple explanation of how catalytic converters work on your car.
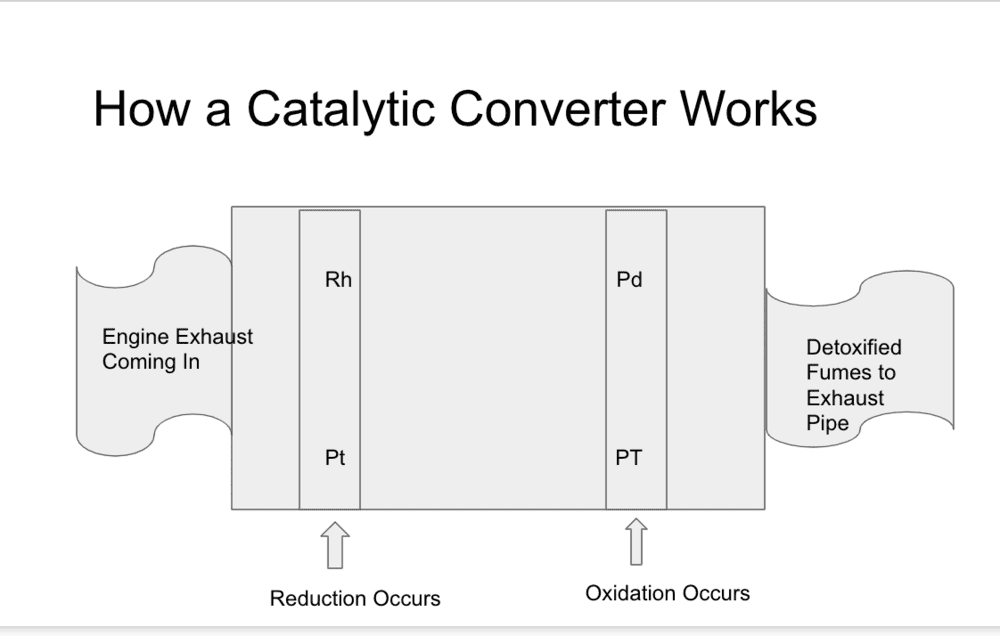
(Pic 1. Process Flow for engine exhaust through the Catalytic Converter)
First Off, Why Do We Have Catalytic Converters?
The US has made catalytic converters mandatory on every car since 1975. Catalytic converters are the part of the car’s exhaust system that cleans the harmful emissions from your car’s engine before the exhaust is released into the air. The converter is located at the bottom of your vehicle just behind the engine.
What’s the importance of recycling them?
Catalytic Converters have value for recycling because they contain precious metals Rhodium (Rh), Platinum (Pt), and Palladium (Pd). These precious metals play an important role in the vehicle’s emission system used to meet the US EPA standards. The precious metals act as “catalysts” to the harmful emission fumes. Then those harmful emission fumes are “converted”, thus leading to the name “Catalytic Converters”.
How Catalytic Converters Work – A Step-by-Step Process
- The process as seen in Pic 1 above is the engine exhaust fumes enter the converter and are first filtered by the Platinum (Pt) and Rhodium (Rh) which is the reduction phase.
- It then moves through the honeycomb to the Oxidation phase this happens with the interaction of Palladium (Pd) and Platinum (Pt).
- The exhaust then exits the catalytic converter and continues through the exhaust pipe.
Watch the video below for a further explanation:
FAIR. CONSISTENT. RELIABLE.
Send your photos to receive a quote today!
GET QUOTE
We at RRCats.com want to thank you for your patronage and for giving us the chance to earn your business & service your Catalytic Converter.